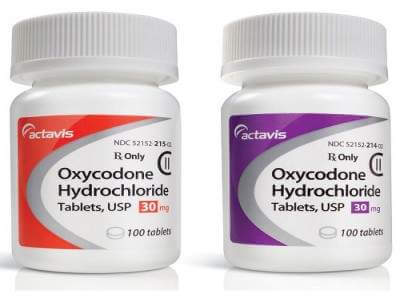
Oxycodone Hydrochloride Tablets
£350.00 – £650.00
Description
oxycodone 30 mg for Sale

Oxycodone hydrochloride tablets are a prescription medication used for the management of moderate to severe pain. Oxycodone is an opioid analgesic that acts on the central nervous system to relieve pain. Here is some information about oxycodone hydrochloride tablets:
1. Pain Relief: They are primarily used to relieve moderate to severe pain. They are often prescribed for pain associated with injuries, surgeries, chronic conditions, or cancer-related pain.
2. Controlled Substance: It classified as a Schedule II controlled substance due to their high potential for abuse and addiction. They should only be used under the supervision and prescription of a healthcare professional.
3. Formulations: They are available in immediate-release (IR) and extended-release (ER) formulations. Immediate-release tablets provide immediate pain relief and are typically taken every 4 to 6 hours as needed. Extended-release tablets provide prolonged pain relief and are taken once or twice daily, depending on the prescribed dosage.
4. Side Effects: Like all opioid medications, oxycodone hydrochloride tablets can cause side effects. Common side effects may include
- drowsiness,
- constipation,
- nausea,
- vomiting,
- dizziness, and respiratory depression.
It is important to report any severe or persistent side effects to a healthcare professional.
5. Potential Risks and Precautions: Oxycodone hydrochloride tablets carry a risk of misuse, abuse, addiction, and overdose. They should not be taken by individuals with a history of substance abuse or addiction. It is important to disclose any medical conditions, medications, or substances being taken to the prescribing healthcare professional to ensure the safe use of oxycodone.
6. Dependency and Withdrawal: Prolonged use or misuse of oxycodone hydrochloride tablets can lead to physical and psychological dependency. Abruptly stopping the medication can result in withdrawal symptoms. It is important to follow the healthcare professional’s guidance for tapering off the medication if discontinuation is necessary.
Dosage and Administration:
The dosage is determined by the healthcare professional based on the individual’s pain severity and response. It is important to follow the prescribed dosage and administration instructions carefully.
Initiate the dosing regimen for each patient individually, taking into account the patient’s severity of pain, patient response, prior analgesic treatment experience, and risk factors for addiction, abuse, and misuse
Monitor patients closely for respiratory depression, especially within the first 24 to 72 hours of initiating therapy and following dosage increases with oxycodone hydrochloride tablets and adjust the dosage accordingly
Patient Access to Naloxone for the Emergency Treatment of Opioid Overdose
Discuss the availability of naloxone for the emergency treatment of opioid overdose with the patient and caregiver and assess the potential need for access to naloxone, both when initiating and renewing treatment with oxycodone hydrochloride tablets
Inform patients and caregivers about the various ways to obtain naloxone as permitted by individual state naloxone dispensing and prescribing requirements or guidelines (e.g., by prescription, directly from a pharmacist, or as part of a community-based program). Consider prescribing naloxone, based on the patient’s risk factors for overdose, such as concomitant use of CNS depressants, a history of opioid use disorder, or prior opioid overdose. The presence of risk factors for overdose should not prevent the proper management of pain in any given patient
Consider prescribing naloxone if the patient has household members (including children) or other close contacts at risk for accidental ingestion or overdose.
Initial Dosage
Although it is not possible to list every condition that is important to the selection of the initial dose of oxycodone hydrochloride tablets, attention must be given to:
1. the daily dose, potency and characteristics of a full agonist or mixed agonist/antagonist the patient has been taking previously.
2. the reliability of the relative potency estimate to calculate the dose of oxycodone HCl needed.
3. the degree of opioid tolerance.
4. the general condition and medical status of the patient, including the patient’s weight and age.
5. the balance between pain management and adverse reactions.
6. the type and severity of the patient’s pain.
7. risk factors for abuse or addiction, including a prior history of abuse or addiction.
Use of Oxycodone Hydrochloride Tablets as the First Opioid Analgesic
Initiate treatment with oxycodone hydrochloride tablets in a dosing range of 5 to 15 mg every 4 to 6 hours as needed for pain. Titrate the dose based upon the individual patient’s response to their initial dose of oxycodone hydrochloride tablets. Patients with chronic pain should have their dosage given on an around-the-clock basis to prevent the reoccurrence of pain rather than treating the pain after it has occurred. This dose can then be adjusted to an acceptable level of analgesia taking into account side effects experienced by the patient.
For control of severe chronic pain, oxycodone hydrochloride tablets should be administered on a regularly scheduled basis, every 4 to 6 hours, at the lowest dosage level that will achieve adequate analgesia.
Conversion from Other Opioids to Oxycodone Hydrochloride Tablets
There is inter-patient variability in the potency of opioid drugs and opioid formulations. Therefore, a conservative approach is advised when determining the total daily dosage of oxycodone hydrochloride tablets. It is safer to underestimate a patient’s 24-hour oxycodone hydrochloride tablets dosage than to overestimate the 24-hour oxycodone hydrochloride tablets dosage and manage an adverse reaction due to overdose. If a patient has been receiving opioid-containing medications prior to taking oxycodone hydrochloride tablets, the potency of the prior opioid relative to oxycodone should be factored into the selection of the total daily dose (TDD) of oxycodone.
In converting patients from other opioids to oxycodone hydrochloride tablets close observation and adjustment of dosage based upon the patient’s response to oxycodone hydrochloride tablets is imperative. Administration of supplemental analgesia for breakthrough or incident pain and titration of the total daily dose of it may be necessary, especially in patients who have disease states that are changing rapidly.
Conversion from Fixed-Ratio Opioid/Acetaminophen, Opioid/Aspirin, or Opioid/Nonsteroidal Combination Drugs
When converting patients from fixed ratio opioid/non-opioid drug regimens a decision should be made whether or not to continue the non-opioid analgesic. If a decision is made to discontinue the use of non-opioid analgesic, it may be necessary to titrate the dose of oxycodone hydrochloride tablets in response to the level of analgesia and adverse effects afforded by the dosing regimen. If the non-opioid regimen is continued as a separate single entity agent, the starting dose oxycodone hydrochloride tablets should be based upon the most recent dose of opioid as a baseline for further titration of oxycodone. Incremental increases should be gauged according to side effects to an acceptable level of analgesia.
Conversion from Oxycodone Hydrochloride Tablets to Extended-Release Oxycodone
The relative bioavailability of oxycodone hydrochloride tablets compared to extended-release oxycodone is unknown, so conversion to extended-release tablets must be accompanied by close observation for signs of excessive sedation and respiratory depression.
Additional information
| Tablets | 100 Tabs, 150 Tabs, 180 Tabs |
|---|



Reviews
There are no reviews yet.